Reverse Osmosis Water Treatment – How RO Filtration Works

What is Reverse Osmosis (RO)?
RO is a filtration technique which uses the particular properties of RO membranes to prevent the passage of a wide range of impurities to a high degree, greatly purifying the feed-water.
What does Reverse Osmosis remove from water?
RO membranes prevent the passage of contaminants in water that are more than about 0.1nm in effective diameter. Typically they remove well over 90% of ionic contamination, most organic compounds and effectively all particulates. RO removal of non-ionic contaminants with molecular weights (MWs) of <100 Dalton can be low. It increases at higher MWs and, in theory, molecules with MWs >300 Dalton, including particles, colloids, microorganisms and large biologically-active molecules, will be rejected completely. Dissolved gases are not removed.
How does Reverse Osmosis work?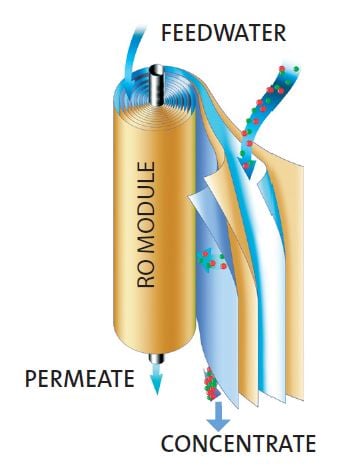
During RO, feed-water is pumped past the input side of an RO membrane under pressure (typically 4-15 bar, 60-220 psi) in cross-flow fashion. Typically 15-30% of feed-water passes through the membrane as permeate and the rest remains on the inlet side and exits as the concentrate stream that contains most of the salts, organics and essentially all particulates.
RO membranes are typically thin film polyamide and are stable over a wide pH range, however, they can be damaged by oxidizing agents such as chlorine, and fouled by organics or colloids, so pre-treatment is usually required to protect the membrane. The membranes require relatively high pressures to overcome back-pressure; to maximise surface area membranes are usually wound in a series of layers in a spiral as shown in the diagram. If the water recovery rate is too high raised concentrations of sparingly soluble hardness salts will lead to membrane blockage.
What are the benefits of Reverse Osmosis?
The great advantage of RO is the very wide range of impurities that it removes. As well as reducing the load on subsequent purification technologies it also protects them from contamination and fouling by large organic molecules, for example, humic and fulvic acids, and completely rejects micro-organisms and viruses present in feed water.
How does ELGA LabWater use Reverse Osmosis?
RO is a key technology in the ELGA PURELAB range. Long lasting, high efficiency membranes are used throughout. ELGA PURELAB stand-alone or pre-treatment units contain RO. Polishers such as those in the Chorus 1 range are designed to ideally be fed with water that has been treated via RO.





