Point of Use Packs fitted to Water Purification Systems
4 Mar 2020

Advantages and Disadvantages of Point of Use Packs fitted to Water Purification Systems
A number of different point-of-use (POU) packs are now available to fit on laboratory water purification systems. These are claimed to reduce the levels of specific types of impurity for particular applications. However, there are major concerns:
- The presence of substantial volumes of reactive and high surface area media in these POU packs after all the water purity monitors means that there is a high likelihood that these devices will contaminate the product water, both initially and after they have been used for some time, with no means of detection.
- For this reason, CLSI (1) and USP (2) specify that the water purity should be monitored as close to the dispense point as possible and, definitely, after all major purification technologies.
- The only technologies accepted in CLSI and USP as point-of-use devices are micro-filters. They are, essentially, physical barriers made from high purity materials and have been found not to degrade the purity of the water as long as they are changed or autoclaved regularly.
- The problem of contamination of product water by POU packs is made much worse by the lack of any water recirculation through them, to keep them clean. Build up of contamination over time is inevitable and it is highly doubtful if the water within the packs would meet CLSI, ASTM and USP standards. Any such contamination would not be detectable.
- The installation of such POU devices, although possibly beneficial for some work, means that the water from the purifier will not always be ultra-pure and necessarily suitable for other applications. The chances of confusion and the inadvertent use of water of an inadequate purity are greatly increased.
To assess if the contamination issues arise in practice, POU packs have been obtained and tested to measure how rapidly they rinse up. The packs were fitted to a water purification system producing ultrapure water with a resistivity of 18.2 MΩ.cm and a TOC of 5ppb. Post-pack resistivity and TOC were monitored on-line.
Multiple tests were carried out on the following types of POU pack:
- A - an ultrafilter used to remove endotoxins and other large biologically-active molecules
- B - a pack containing 50mL activated carbon, used to reduce levels of endocrine disruptors
- C - a pack containing 25g C18 reverse-phase media, used to remove trace contaminants from water for HPLC
- D - a pack containing 50mL activated carbon, used to remove trace volatile organic compounds (VOC)
- E - An ELGA Biofilter (a positively charged POU micro-filter used to remove bacteria and endotoxin.
- F - a 0.2µm POU micro-filter used to remove bacteria
The results are summarised in the table below. Prior to the rinse tests, pack C was flushed with 50mL methanol and then with ultrapure water for 10 minutes. Typical rinse profiles for resistivity and TOC for the various packs are shown in figures 1 and 2.
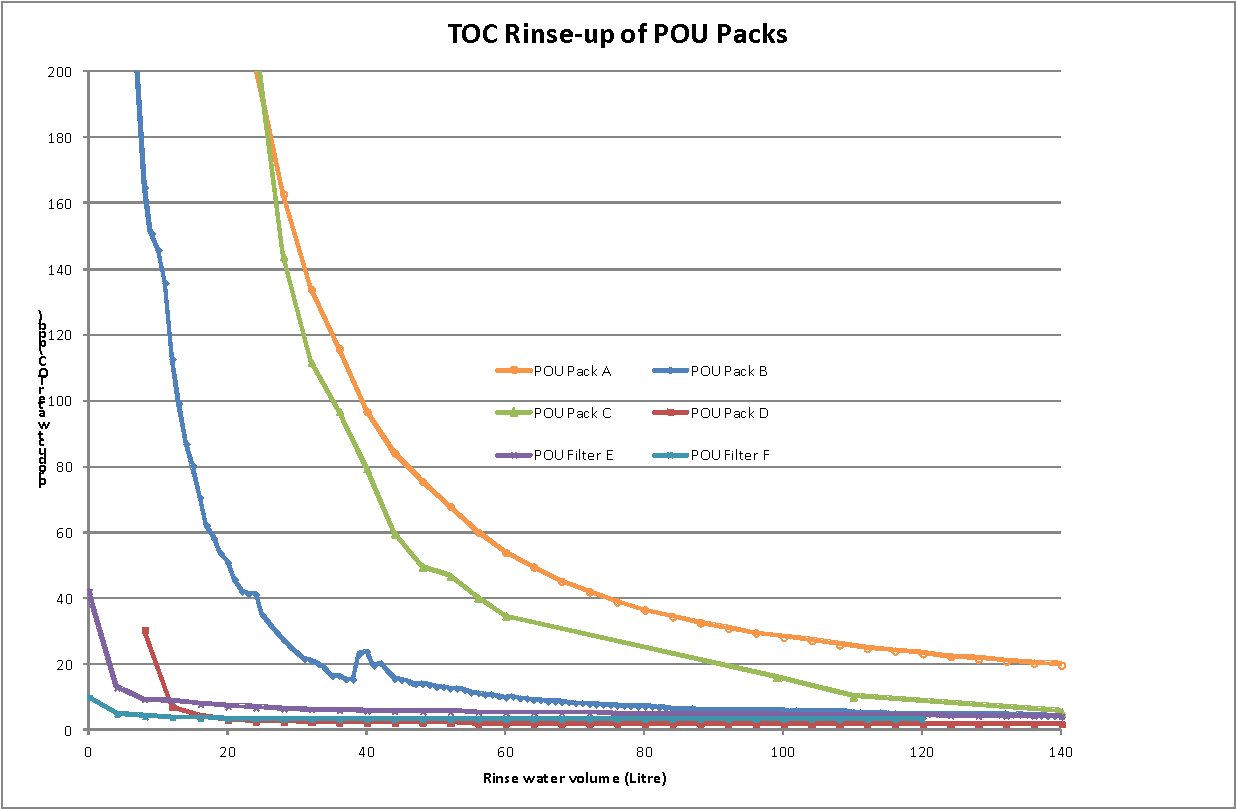
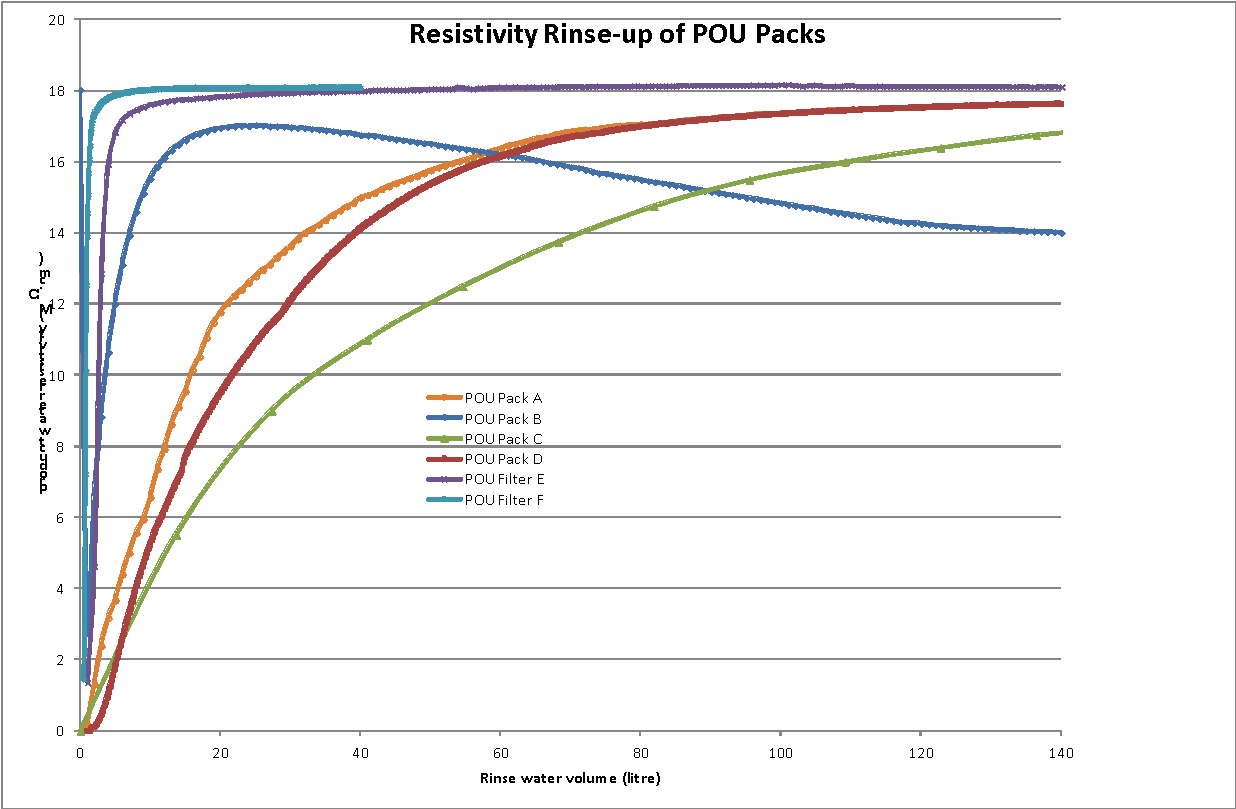
The POU packs showed varying rates of rinse-up. Packs A, B , C, and D all required over 100 liters of water to achieve a product water resistivity of >17.5 MΩ.cm. Pack B was unusual as it consistently rinsed fairly rapidly initially and then started to release further ionic contaminants; it did not reach 17.5 MΩ.cm even after 4000 liters! TOC rinse up was rather more consistent but packs A, B and C took over 100 liters to reach less than 10ppb. Pack A was particularly slow and the initial rinse water contained a TOC over 5000ppb. In contrast, pack F, the 0.2µm micro-filter rinsed rapidly for both resistivity and TOC, as did pack E, the ELGA Biofilter.
The work described here validates the practice of fitting only micro-filters as POU devices in laboratory water purification systems. Installing any POU packs on the outlets of the system dispenser, long after any monitoring, must be viewed as a retrograde step both in principle for reasons outlined above and, as shown in practice, from the experimental results. Such POU treatments introduce a large volume of reactive and high surface area media after all monitoring has taken place. In these situations, there cannot be validatable control of product water purity.
References
- Clinical and Laboratory Standards Institute. Preparation and Testing of Reagent Water in the Clinical Laboratory; Approved Guideline – 4th Edition. Page 5.
- US Pharmacopeia
Dr Paul Whitehead
After a BA in Chemistry at Oxford University, Paul focused his career on industrial applications of chemistry. He was awarded a PhD at Imperial College, London for developing a microwave-induced-plasma detector for gas chromatography. He spent the first half of his career managing the analytical support team at the Johnson Matthey Research/Technology Centre,specialising in the determination of precious metals and characterising applications such as car-exhaust catalysts and fuel cells. Subsequently, as Laboratory Manager in R&D for ELGA LabWater, he has been involved in introducing and developing the latest water purification technologies. He now acts as a consultant for ELGA.
